![Exceptional case, Hybridisation ,shape of an outer orbital complex [ Ni(NH3)6]2+, Octahedral complex - YouTube Exceptional case, Hybridisation ,shape of an outer orbital complex [ Ni(NH3)6]2+, Octahedral complex - YouTube](https://i.ytimg.com/vi/R5RDFu1oYUU/maxresdefault.jpg)
Exceptional case, Hybridisation ,shape of an outer orbital complex [ Ni(NH3)6]2+, Octahedral complex - YouTube
2 – topic of research paper in Chemical sciences. Download scholarly article PDF and read for free on CyberLeninka open science hub. Thermal decomposition of polycrystalline [Ni(NH3)6](NO3)2 – topic of research paper in Chemical sciences. Download scholarly article PDF and read for free on CyberLeninka open science hub.](https://cyberleninka.org/viewer_images/173973/f/1.png)
Thermal decomposition of polycrystalline [Ni(NH3)6](NO3)2 – topic of research paper in Chemical sciences. Download scholarly article PDF and read for free on CyberLeninka open science hub.
![Explain the hybridisation, magnetic property and geometry [Ni(CN)4]2− and [ Ni(NH3)4]2+ using VB theory. Explain the hybridisation, magnetic property and geometry [Ni(CN)4]2− and [ Ni(NH3)4]2+ using VB theory.](https://search-static.byjusweb.com/question-images/toppr_ext/questions/633645_607318_ans_06ef044c85004ad2aed957fc1fd1f24d.png)
Explain the hybridisation, magnetic property and geometry [Ni(CN)4]2− and [ Ni(NH3)4]2+ using VB theory.
![Why is [Ni(NH3)6]Cl2 paramagnetic but [Co(NH3)6]Cl3 is diamagnetic ? | Electron configuration, Coordination number, Crystal field theory Why is [Ni(NH3)6]Cl2 paramagnetic but [Co(NH3)6]Cl3 is diamagnetic ? | Electron configuration, Coordination number, Crystal field theory](https://i.pinimg.com/736x/9e/ad/9f/9ead9f774d9995ac47c672b48098f159.jpg)
Why is [Ni(NH3)6]Cl2 paramagnetic but [Co(NH3)6]Cl3 is diamagnetic ? | Electron configuration, Coordination number, Crystal field theory

Inorganics | Free Full-Text | Ni(NH3)2(NO3)2—A 3-D Network through Bridging Nitrate Units Isolated from the Thermal Decomposition of Nickel Hexammine Dinitrate
![why [Ni(NH3)4]2+ has tetrahedral geometry Explain with vbt - Chemistry - Coordination Compounds - 9638641 | Meritnation.com why [Ni(NH3)4]2+ has tetrahedral geometry Explain with vbt - Chemistry - Coordination Compounds - 9638641 | Meritnation.com](https://s3mn.mnimgs.com/img/shared/ck-files/ck_56065adb7f230.png)
why [Ni(NH3)4]2+ has tetrahedral geometry Explain with vbt - Chemistry - Coordination Compounds - 9638641 | Meritnation.com
![21 15. The oxidation state of Ni and NH, in [Ni(NH3)4]+2 (1) Ni = +2, NH, = 0 [RPMT 2001 (2) Ni = +1, NH, = - 1/6 (3) Ni = +1, NH, = + 1/6 (4) Ni = 0, NH, = +2 21 15. The oxidation state of Ni and NH, in [Ni(NH3)4]+2 (1) Ni = +2, NH, = 0 [RPMT 2001 (2) Ni = +1, NH, = - 1/6 (3) Ni = +1, NH, = + 1/6 (4) Ni = 0, NH, = +2](https://toppr-doubts-media.s3.amazonaws.com/images/4816097/e84878e9-2fe2-45ee-a89d-cd525e6ad65a.jpg)



![why cant [Ni(NH3)6]^2+ have dsp3 hybridization then? : r/CBSE why cant [Ni(NH3)6]^2+ have dsp3 hybridization then? : r/CBSE](https://preview.redd.it/why-cant-ni-nh3-6-2-have-dsp3-hybridization-then-v0-slwfvbv55ria1.png?width=640&crop=smart&auto=webp&s=f4e658df50fc25da185b4ccd9590bfb69fc4c902)

2. The... | Download Scientific Diagram Representation of the cubic structure of [Ni(NH3)6](NO3)2. The... | Download Scientific Diagram](https://www.researchgate.net/publication/269400063/figure/fig8/AS:667921089036294@1536256205990/Representation-of-the-cubic-structure-of-NiNH36NO32-The-constituent-atoms.jpg)
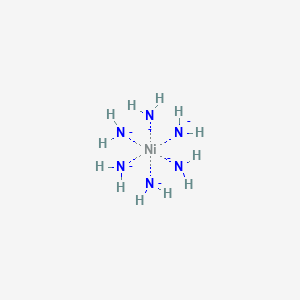




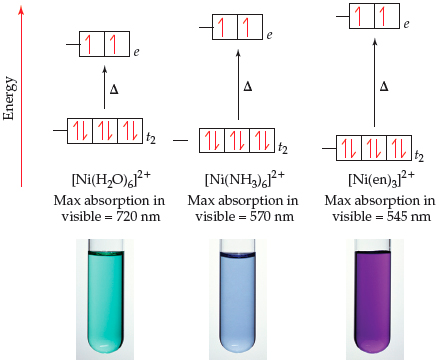

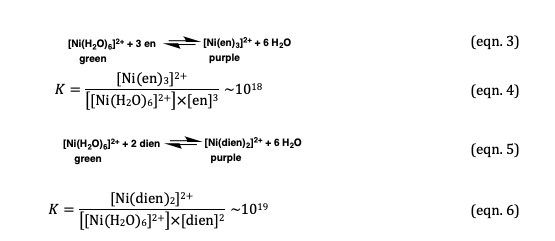


![How is [Ni(NH3) 6] 2+ paramagnetic while [Ni(CN) 6] 4- diamagnetic? - Quora How is [Ni(NH3) 6] 2+ paramagnetic while [Ni(CN) 6] 4- diamagnetic? - Quora](https://qph.cf2.quoracdn.net/main-qimg-f7fd36508df0305723aa4956637d3097.webp)