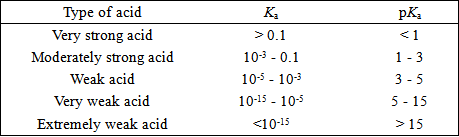
For a weak monobasic acid, if pK4 = 4 , then at a concentration of 0.01 M of the acid solution, the van't Hoff factor is:
![PDF] The Influence of Ionic Strength on Apparent and Thermodynamic Parameters ( Ka , pKa ' s ) for HF and Phosphate Buffer Capacities | Semantic Scholar PDF] The Influence of Ionic Strength on Apparent and Thermodynamic Parameters ( Ka , pKa ' s ) for HF and Phosphate Buffer Capacities | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/c0a6140f88db06398f56b28ce5fd6ac92cd82d39/4-Table1-1.png)
PDF] The Influence of Ionic Strength on Apparent and Thermodynamic Parameters ( Ka , pKa ' s ) for HF and Phosphate Buffer Capacities | Semantic Scholar





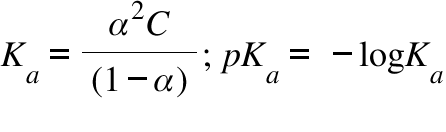



:max_bytes(150000):strip_icc()/what-is-pka-in-chemistry-605521_FINAL2-9fdfc39e9aa34caa96d6e74a2c687707.png)